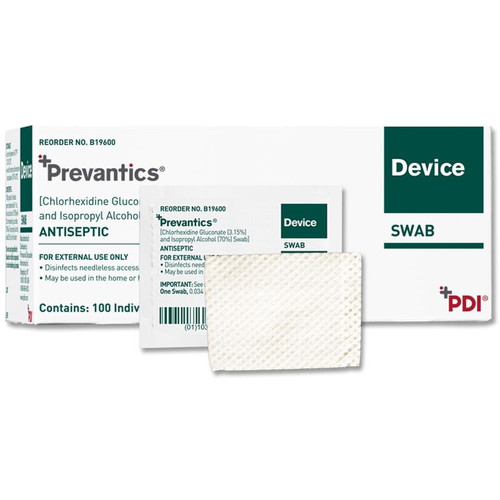
Disinfect needleless access sites prior to use by scrubbing the hub with Prevantics Device Swab. This swab contains 3.15% (w/v) Chlorhexidine gluconate and 70% (v/v) Isopropyl alcohol solution.
Prevantics Device SwabFeatures:
Intuitive prep-pad design 1mL 100 swabs/box Scrub and Dry Time
5 second scrub time followed by 5 second dry time Formulation
3.15% (w/v) Chlorhexidine gluconate (CHG) and 70% (v/v) Isopropyl alcohol (IPA) Applicator
Intuitive, easy-to-use prep pad format provides friction to remove microorganisms from grooves and threads of connectors Efficacy
Achieved a >4.0 log10 reduction versus a leading alcohol impregnated cap1 against 7 tested clinically relevant microorganisms known to cause CLABSIs2 Guideline Recommended
Effectively scrub the hub prior to and between each line access, as recommended in the Infusion Nurse Society (INS) Guidelines FDA Cleared
First and only 3.15% CHG/70% IPA swab to receive 510(k) clearance from the US Food and Drug Administration (FDA) for disinfecting needleless access sites prior to use Use Sites
Disinfecting needleless access sites (needleless connectors, injection ports, and access ports) and tops of blood culture collection bottles. Independent Data
Over 15 independent clinical studies demonstrating clinical efficacy of Prevantics products Compliance
US Centers for Disease Control and Prevention Infusion Nurses Society Society for Healthcare Epidemiology of America The Joint Commission National Patient Safety Goals Infectious Diseases Society of America Association for Professionals in Infection Control and Epidemiology Association for Vascular Access